Update: Clinical trials
Jenny Townhill and Tim McLean, Central Coordination
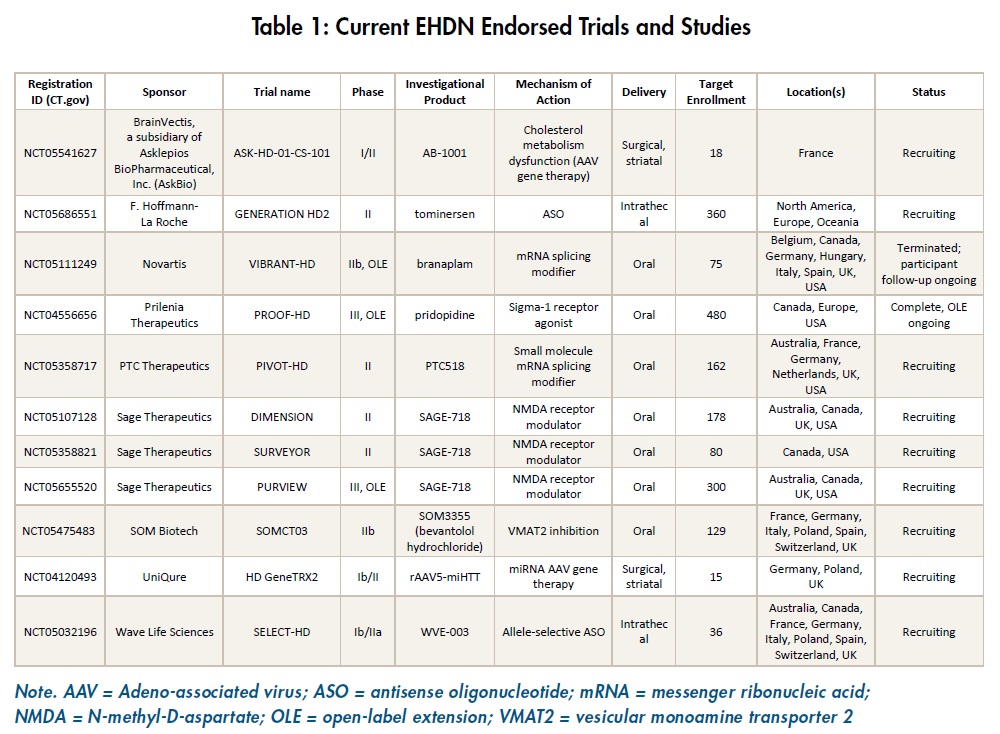
The following studies have been endorsed by EHDN. Endorsement of a study protocol follows review by the EHDN Scientific and Bioethics Advisory Committee, which makes its recommendations to the Executive Committee. If endorsed, a formal letter of endorsement is then issued to the study sponsor, allowing them to inform relevant regulatory authorities and/or ethics committees that the study protocol has been reviewed and endorsed by a group of expert HD scientists and clinicians. The endorsement may also be posted on the EHDN website and shared on social media, signalling the same message to the HD community.
The studies reported below are of investigational compounds, where safety and efficacy have not been established. There is no guarantee that the outcome of these studies will result in marketing approval.
Key updates since the last newsletter are provided below for EHDN-endorsed trials and studies; please refer to Table 1 for a summary of the main study information.
 ASK-HD-01-CS-101 (Asklepios Biopharmaceutical, Inc. [AskBio])
ASK-HD-01-CS-101 (Asklepios Biopharmaceutical, Inc. [AskBio])
This surgical open-label phase I/II study evaluating the safety and efficacy of BV-101, a novel gene therapy targeting cholesterol metabolism dysfunction opened to recruitment in Q42022. Up to 18 participants will be enrolled at two sites in France.
![]() VIBRANT-HD (Novartis)
VIBRANT-HD (Novartis)
This study was designed to detect and assess early safety signals in the development of branaplam in the treatment of HD. In December 2022, Novartis shared the news there will be no further dosing of branaplam in the VIBRANT-HD study and that the development of branaplam for HD will not continue. The study had previously been paused following the detection of side effects in some participants. A review of additional follow-up data showed that many participants who received branaplam had signs or symptoms of peripheral neuropathy. There was also an increase in a marker of neuronal damage [neurofilament light chain (NfL)] and some MRI findings suggesting an increase in the volume of the lateral ventricles.
Branaplam did lower mutant huntingtin protein (mHTT) in the cerebral spinal fluid as expected. However, given the safety findings, it is very unlikely that it is safe to explore other doses or other dosing frequencies.
All participants who received the study drug will continue to be followed up for one year after their last dose of branaplam to monitor recovery and gather more data about these safety signals and how they may change over time.
 DIMENSION, SURVEYOR and PURVIEW (Sage Therapeutics)
DIMENSION, SURVEYOR and PURVIEW (Sage Therapeutics)
Recruitment is continuing into the two phase II trials, DIMENSION and SURVEYOR, assessing the effect of an orally administered NMDA receptor modulator, SAGE-718, on cognitive performance and functioning. A further phase III open-label study of SAGE-718, PURVIEW, has started recruitment and will enrol a total of 300 participants, comprising participants from DIMENSION and SURVEYOR as well as an additional cohort of de novo participants who were not enrolled in the phase II studies. The PURVIEW trial has been granted partial endorsement; the transition of DIMENSION and SURVEYOR participants to PURVIEW has been endorsed, however, as the de novo cohort has no placebo arm, and no data currently exists to provide appropriate matching of participants, this additional cohort has not been endorsed.
 HDGeneTRX1 and HDGeneTRX2 (uniQure)
HDGeneTRX1 and HDGeneTRX2 (uniQure)
Last summer, unexpected severe side effects, including localised inflammatory responses, that were suspected to be related to the study drug were reported by uniQure for some participants that were treated with the higher dose of AMT-130. At the time, the trial data safety monitoring board (DSMB) recommended temporarily delaying further enrolment into this higher-dose group pending a full safety review. Following this review of safety data, the trial DSMB determined that it was safe to proceed with recruitment into the higher dose group and additional safety measures, including closer post-surgical monitoring, have been included in the assessment protocol.
 PIVOT-HD (PTC Therapeutics)
PIVOT-HD (PTC Therapeutics)
Recruitment continues into this 12-month phase IIa study of orally administered PTC518 (a small molecule huntingtin-lowering compound) in Australia and Europe. Currently, the study is on hold for sites in the US as the FDA has requested additional data to allow the study to proceed. Preliminary data are expected to be reported early in 2023.
 GENERATION HD2 (F. Hoffman La Roche)
GENERATION HD2 (F. Hoffman La Roche)
This phase II study of intrathecally administered tominersen, a huntingtin-lowering antisense oligonucleotide (ASO), will evaluate the safety, biomarkers and efficacy trends of different dose levels of tominersen in people with prodromal
(roughly equivalent to HD-ISS Stage 2) or early manifest HD (roughly equivalent to HD-ISS Stage 3). Approximately 360 participants will be enrolled across 15 countries, and each participant will have at least 16 months of treatment with either tominersen or placebo.
The study has started screening, with the first sites activated in the US and Spain at the start of 2023, and additional sites in North America, Europe, South America and Oceania expected to open in the coming months. Details will be posted on clinicaltrials.gov as individual sites are activated and are ready to start screening potential participants.
 SELECT-HD (Wave Life Sciences)
SELECT-HD (Wave Life Sciences)
This phase Ib/IIa study of an allele-selective ASO continues to recruit participants. Preliminary data were reported last year on the first two of the three single-dose cohorts, showing that a single dose of WVE-003 reduces mHTT in cerebrospinal fluid (CSF) and that WVE-003 was in general, safe and well-tolerated. Normal htt levels were consistent with allele selectivity. There is no apparent difference in dose response between the two dose groups analysed to date. Based on these findings, the originally planned number of participants that will be included in each single dose cohort has been increased to gather additional data on dose response and the percentage reduction of mHTT in CSF. Additional bio-marker and safety data for the single-dose cohorts are expected in the first half of 2023.
 SHIELD-HD (Triplet Therapeutics)
SHIELD-HD (Triplet Therapeutics)
This study is now closed. Seventy prodromal and early manifest HD gene expansion carriers were enrolled in this 96-week natural history study, and the last 96-week visit was completed towards the end of 2022. The primary objectives of the study were to assess DNA damage repair (DDR) gene expression, and somatic instability (SI) of the HD gene and to examine associations between DDR gene expression, SI and markers of disease progression which included validated and novel clinical outcome measures, wet biomarkers such as NfL, and neuroimaging.